Get fastest alerts on Results, Admit Cards & Govt Jobs directly on your phone.
India sits at a strange crossroads — it is one of the world’s largest biotech economies, yet it has approved only one genetically modified crop for commercial cultivation. That single fact tells you everything about how regulation, science, and politics collide in Indian biotechnology policy. If you are preparing for GS-III, understanding this regulatory maze is not optional. UPSC has tested it repeatedly, and the questions are getting sharper every cycle.
I have spent years helping aspirants decode science and technology topics for Mains, and I can tell you that most students make one mistake here. They memorise the names of committees and acts but never understand how the system actually works. This article will fix that. We will go from the legal foundation all the way to the policy debates that show up in 200-word answers.
Where This Topic Sits in the UPSC Syllabus
Biotechnology regulation falls squarely under GS-III. The syllabus mentions “Science and Technology — developments and their applications and effects in everyday life” and “Awareness in the fields of IT, Space, Computers, Robotics, Nano-technology, Bio-technology.” Policy-level questions also connect to the “Conservation, Environmental Pollution and Degradation” segment because GM organisms raise biosafety concerns.
| Exam Stage | Paper | Syllabus Section |
|---|---|---|
| Prelims | General Studies | Science and Technology, Environment and Ecology |
| Mains | GS-III | Science and Technology: Developments and Applications; Conservation and Biosafety |
| Mains | GS-II | Government Policies and Interventions (when regulation angle is tested) |
Questions on this topic have appeared in Prelims at least 5-6 times since 2010, often as factual questions about GEAC, Bt crops, or biosafety protocols. In Mains, the examiner prefers policy analysis — asking you to evaluate whether India’s regulatory framework is adequate for the pace of biotech innovation.
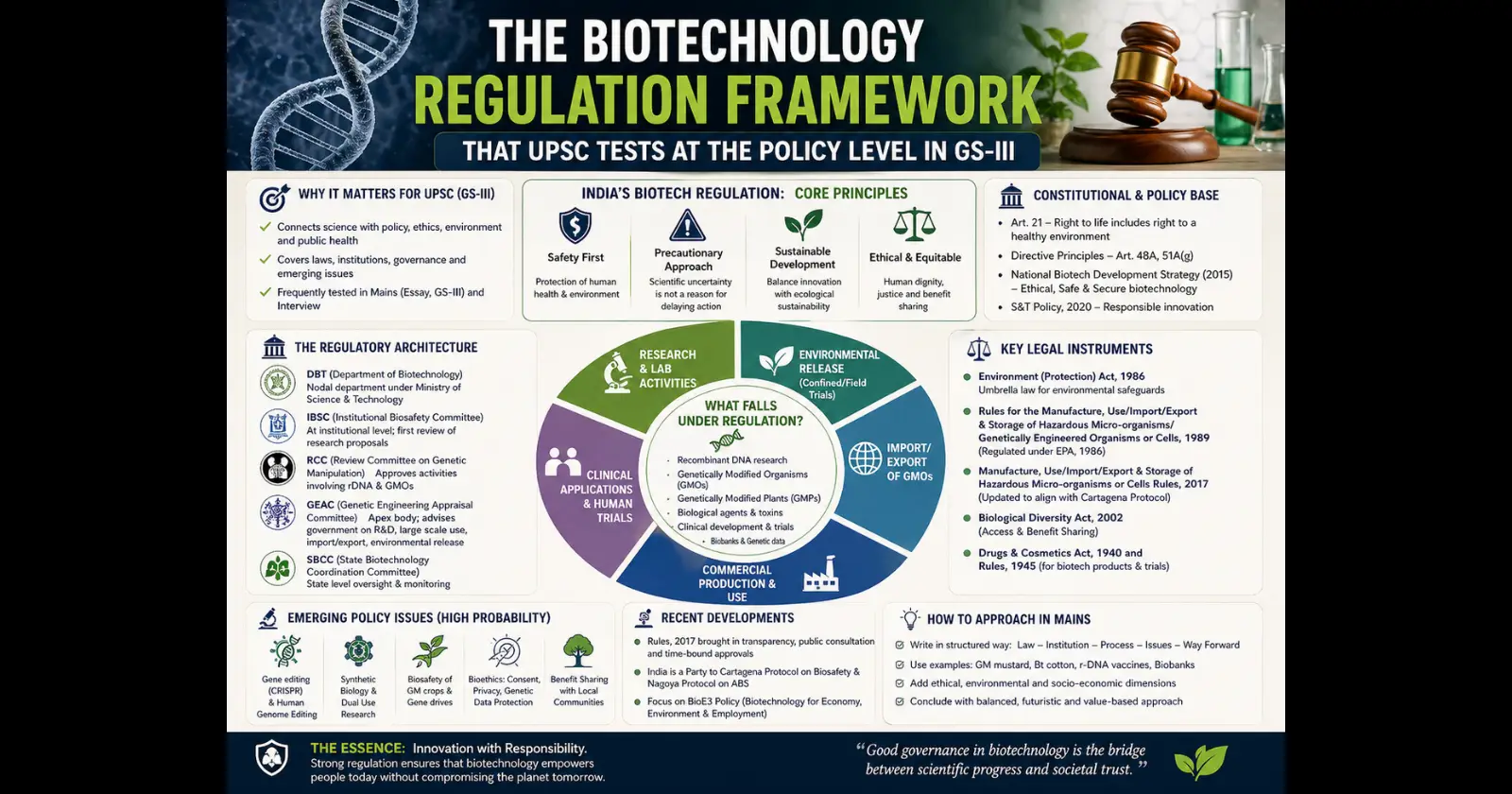
The Legal Foundation — Where It All Begins
India does not have a standalone biotechnology regulation law. That surprises many aspirants. Instead, the entire framework rests on the Environment Protection Act (EPA), 1986. Under this Act, the government notified the Rules for the Manufacture, Use, Import, Export and Storage of Hazardous Microorganisms/Genetically Engineered Organisms or Cells, 1989. These are commonly called the Rules 1989.
Think of it this way. The EPA 1986 is the parent law. The Rules 1989 are the specific instructions for handling anything genetically modified. Every committee, every approval process, every safety assessment traces back to these two documents.
The Rules 1989 created a multi-layered system of committees. Each committee operates at a different level — from the institutional lab all the way up to the national government. This layered approach was designed to ensure that no single authority could push through an approval without checks.
The Regulatory Bodies — Who Does What
There are six statutory committees under the Rules 1989. For UPSC, you need to know three of them well and be aware of the others.
The Institutional Biosafety Committee (IBSC) operates at the level of every research institution or company working with GMOs. It is the first checkpoint. Any scientist wanting to conduct experiments with genetically engineered organisms must get IBSC approval first. The institution itself sets up this committee, but it must include a Department of Biotechnology (DBT) nominee.
The Review Committee on Genetic Manipulation (RCGM) functions under the DBT. It monitors ongoing research, reviews safety data from lab and field trials, and gives permission for small-scale field trials. Think of RCGM as the technical gatekeeper — it evaluates the science before anything reaches the final decision-makers.
The Genetic Engineering Appraisal Committee (GEAC) is the apex body. It functions under the Ministry of Environment, Forest and Climate Change (MoEFCC). GEAC gives the final approval for large-scale field trials and commercial release of any genetically modified organism in India. No GM crop can be sold to farmers without GEAC clearance. This is the body that approved Bt Cotton in 2002 and that has been at the centre of the Bt Brinjal controversy since 2010.
One detail that UPSC loves to test: GEAC was originally called the Genetic Engineering Approval Committee. It was renamed to Genetic Engineering Appraisal Committee to signal that its role is assessment, not just rubber-stamping approvals.
The Bt Cotton Story and the Bt Brinjal Moratorium
Bt Cotton remains the only GM crop approved for commercial cultivation in India. It was approved in 2002 after years of field trials. The Bt gene (from the bacterium Bacillus thuringiensis) produces a protein toxic to bollworm, a major cotton pest. India’s cotton production did increase significantly after Bt Cotton adoption, though debates continue about farmer dependency on seed companies and the emergence of pest resistance.
In 2009, GEAC approved Bt Brinjal for commercial release. However, in February 2010, the then Environment Minister imposed an indefinite moratorium on Bt Brinjal after conducting public consultations across the country. The moratorium cited the need for more independent safety testing and the precautionary principle. As of 2026, that moratorium has not been lifted. This case is a textbook example of how science, public opinion, and political decision-making interact in Indian policy — exactly the kind of analysis UPSC expects in a Mains answer.
The International Dimension — Cartagena Protocol
India is a party to the Cartagena Protocol on Biosafety, which operates under the Convention on Biological Diversity (CBD). The Protocol governs the transboundary movement of living modified organisms (LMOs). It enshrines the precautionary approach — meaning a country can refuse entry of a GMO even without full scientific certainty of harm.
For UPSC, the connection is direct. India’s domestic moratorium on Bt Brinjal aligns with the precautionary principle embedded in the Cartagena Protocol. When you write a Mains answer, linking domestic policy to international commitments strengthens your analysis considerably.
The Gaps in the Current Framework
The regulatory system has well-known weaknesses that UPSC has probed in past papers. First, there is no dedicated biotechnology regulatory law. The Rules 1989 were framed when the biotech sector barely existed in India. Technologies like gene editing (CRISPR-Cas9), synthetic biology, and gene drives do not fit neatly into a framework designed for first-generation GM crops.
Second, there is an inherent conflict of interest. The DBT both promotes biotechnology research and regulates it through RCGM. This is like asking a department to be both cheerleader and referee. The proposed Biotechnology Regulatory Authority of India (BRAI) Bill was introduced in 2013 to create an independent, statutory regulator. It lapsed and has not been reintroduced.
Third, state governments have sometimes resisted GEAC decisions. Several states have refused to allow field trials of GM crops on their soil, raising questions about Centre-State coordination on biosafety — a federalism angle that connects to GS-II as well.
Recent Policy Developments
The National Biotechnology Development Strategy 2021-25 laid out India’s vision for making the biotech sector a key economic driver. It emphasised regulatory reform, ease of doing research, and building biosafety capacity. The DBT has also issued guidelines for genome-edited organisms, attempting to distinguish between transgenic organisms (foreign gene inserted) and gene-edited organisms (small changes within the organism’s own genome). This distinction matters because many countries regulate gene-edited crops less strictly than transgenic ones.
In 2024, India permitted environmental release of certain gene-edited plants that do not contain foreign DNA, signalling a possible shift in regulatory philosophy. Watch this space — UPSC is likely to frame questions around whether India’s regulatory approach should differentiate between gene editing and traditional genetic modification.
Previous Year UPSC Questions on This Topic
Q1. What are the Genetically Modified crops? Discuss the issues related to their cultivation in India.
(UPSC Mains 2019 — GS-III, 15 marks)
Answer: Genetically modified crops are plants whose DNA has been altered using genetic engineering to introduce a desired trait — such as pest resistance, drought tolerance, or improved nutrition. In India, Bt Cotton is the only approved GM crop. The issues around GM cultivation include biosafety concerns about long-term ecological impact, farmer dependency on proprietary seeds, inadequacy of the regulatory framework under the Rules 1989, the unresolved Bt Brinjal moratorium, Centre-State disagreements on field trials, and the absence of a standalone biotech regulator like the proposed BRAI. Supporters argue GM crops can boost productivity and reduce pesticide use, while critics invoke the precautionary principle and demand more independent testing. India needs a balanced, science-based regulatory approach that protects both farmers and the environment.
Explanation: This question tested whether aspirants could go beyond definitions and discuss the policy, economic, and environmental dimensions of GM crops. The examiner wanted a structured answer covering what GM crops are, India’s experience with them, and the multi-dimensional debate. Linking to the Cartagena Protocol and the BRAI Bill would earn extra credit.
Q2. Which of the following statements about GEAC is/are correct?
1. It functions under the Ministry of Science and Technology.
2. It gives approval for commercial release of GM organisms.
(UPSC Prelims 2017 — General Studies)
Answer: Only statement 2 is correct. GEAC functions under the Ministry of Environment, Forest and Climate Change, not the Ministry of Science and Technology. Many aspirants confuse this because the DBT (which houses RCGM) is under the Ministry of Science and Technology. GEAC and RCGM are under different ministries — this is a favourite trap in Prelims.
Q3. Discuss the need for an independent biotechnology regulator in India. Examine the key features and fate of the BRAI Bill.
(UPSC Mains 2020 — GS-III, 10 marks)
Answer: India’s biotech regulation currently operates under the EPA 1986 and Rules 1989, with multiple committees across different ministries. The lack of a single, independent regulator creates overlap, delays, and conflict of interest — particularly since DBT both promotes and regulates biotech. The BRAI Bill 2013 proposed an independent statutory authority to streamline approvals, ensure biosafety, and build public trust. However, it faced opposition from environmental groups who felt it prioritised industry over safety, and from states concerned about losing their say. The Bill lapsed with the dissolution of the 15th Lok Sabha and has not been reintroduced. An independent regulator remains a critical gap in India’s biotech governance.
Explanation: This question directly tested awareness of institutional reform in science governance. The examiner wanted aspirants to identify the structural problem (no independent regulator), explain the proposed solution (BRAI), and analyse why it failed politically. This is a classic GS-III governance-meets-science question.
Key Points to Remember for UPSC
- India’s biotech regulation rests on the EPA 1986 and Rules 1989 — there is no standalone biotech law.
- GEAC (under MoEFCC) is the apex body for commercial release of GMOs; RCGM (under DBT) handles research-stage approvals.
- Bt Cotton (approved 2002) is the only commercially cultivated GM crop in India; Bt Brinjal has been under moratorium since 2010.
- The BRAI Bill 2013 proposed an independent regulator but lapsed and has not been revived.
- India is a signatory to the Cartagena Protocol on Biosafety, which supports the precautionary approach to GMOs.
- The DBT’s dual role as promoter and regulator of biotech is a structural conflict of interest — a point worth raising in Mains answers.
- Gene editing technologies like CRISPR are pushing India to update its regulatory categories beyond traditional transgenics.
- Centre-State friction over GM field trials adds a federalism dimension that can connect your answer to GS-II as well.
Understanding India’s biotech regulatory framework gives you material for questions across GS-II and GS-III — from governance and federalism to science policy and environmental safety. As a next step, read the original text of the Rules 1989 (it is short and available on the MoEFCC website) and make a one-page flowchart of how a GM crop moves from lab to field to market. That single exercise will make this topic stick far better than any amount of passive reading.